

Boosting electrochemical conversion using a pulsating flow electrolyzer
24-06-2020 | Research result
 In a recent publication in the journal Industrial & Engineering Chemical Research, VoltaChem researchers demonstrate how a pulsating flow can boost electrochemical conversion without the need for a complex reactor design. As a test reaction, the researchers investigated the conversion of 1,2-propanediol (PDO) to lactic acid (LA) and further to pyruvic acid (PA). The pulsation flow electrolyzer not only helps to overcome mass transport limitation, it can also enhance the selectivity of the conversion.
In a recent publication in the journal Industrial & Engineering Chemical Research, VoltaChem researchers demonstrate how a pulsating flow can boost electrochemical conversion without the need for a complex reactor design. As a test reaction, the researchers investigated the conversion of 1,2-propanediol (PDO) to lactic acid (LA) and further to pyruvic acid (PA). The pulsation flow electrolyzer not only helps to overcome mass transport limitation, it can also enhance the selectivity of the conversion.
Electrosynthesis is a promising technology for the transition toward a carbon-neutral chemical industry. However, not many electrochemical conversions are already performed on an industrially relevant scale. The main challenge is the mass transport limitation of most reactions, often caused by low solubility of reactants (in particular organic compounds) in the electrochemical reaction medium. This results in low productivity and energy efficiency and often limits the selectivity toward the desired product.
A novel approach in electrochemistry
As a solution to this challenge, the VoltaChem researchers propose the use of a pulsed flow reactor (pictured above). It can enhance mass transfer without the need of a complex reactor design. Although the pulsating strategy is common for processes such as extraction and emulsion polymerization, this is a novel approach for electrochemical processes.
In their publication in Industrial & Engineering Chemical Research, the researchers describe the application of a pulsed electrolyte flow in an electrochemical flow cell. They used the conversion of 1,2-propanediol (PDO) to lactic acid (LA) and further to pyruvic acid (PA) as a test reaction. This reaction was selected for its economic potential: lactic acid can be used as a building block for biodegradable polymers.
To assess the performance of the pulsed flow electrolysis, the researchers performed a parametric study of flowrates, pulsation frequency, and amplitude in the different compartments of the electrolyzer. In addition, they developed a phenomenological mathematical model to investigate the effect of the pulsed flow on the mass transfer.
Increased yield combined with higher selectivity
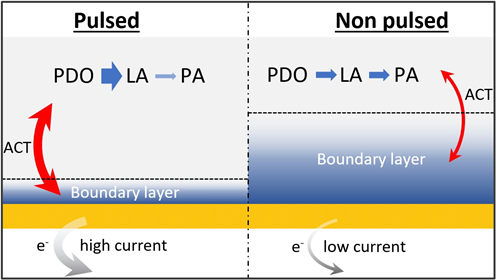 The research indeed showed an improved mass transfer of electroactive species due to a higher instantaneous velocity driven by the pulsations. Though the net residence time of the reactants was not altered by the pulsation, the enhancement of mass transfer leads to an increase of the conversion. The pulsating regime was shown to result in a yield increase of lactic acid of a factor of two.
The research indeed showed an improved mass transfer of electroactive species due to a higher instantaneous velocity driven by the pulsations. Though the net residence time of the reactants was not altered by the pulsation, the enhancement of mass transfer leads to an increase of the conversion. The pulsating regime was shown to result in a yield increase of lactic acid of a factor of two.
More importantly it was established that the selectivity for the products of PDO oxidation (LA and PA) can be tuned by adjusting the flowrates and the pulse frequency of the electrolytes for a given displacement volume. Thus a 15-20% gain in selectivity could be achieved, to a total of 95% towards lactic acid.
Paper
Elena Pérez-Gallent, Carlos Sánchez-Martínez, Leon F. G. Geers, Susan Turk, Roman Latsuzbaia, and Earl L. V. Goetheer: Overcoming Mass Transport Limitations in Electrochemical Reactors with a Pulsating Flow Electrolyzer.Ind. Eng. Chem. Res. 2020, 59, 5648−5656 DOI: 10.1021/acs.iecr.9b06925
Would you like to learn more about different approaches to boost electrochemical conversion? Contact Earl Goetheer, technical lead of the VoltaChem Power-2-Chemicals program line.
Share this page: